Why water is liquid at room temperature? Water can be liquid in room temperatures. If we put it in freezer, water will freeze, become solid and turn to ice. If we boil water for our tea or coffee we will notice water vapor. If we boil water long enough we will turn all of our liquid water into water vapor. This is a great examples of three states of matter: SOLID , LIQUID and GASEOUS.
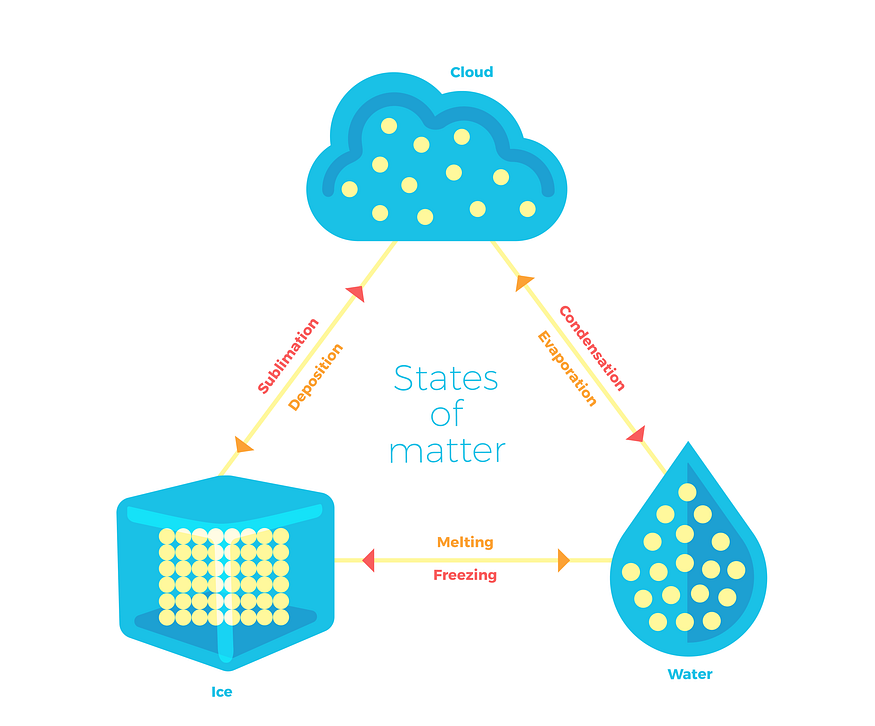
Image from BEAKERLIFE, find ot more on their website.
Water will freeze on 0 ºC, zero degree Celsius and below, and turn from liquid into solid. If we warm it back up it will melt it again to liquid water, and if we warm it even further to 100 ºC it will start boiling and eventually turn into gas. So by increasing the temperature, we can turn water into gas and by decreasing it we can turn it into its solid form or ice.
But this goes not just for water, any substance can have solid, liquid or gaseous state. However some materials like metal are solid already at room temperature and require very high temperatures to be turned into liquid or gas, while some other substance like nitrogen (one of the gases that make air that we breed) require very low temperatures to be turned into liquid (−195.79 ºC) and even lower to be turned into solid.
The reason this happens lies in the structure of each state. In solid state, particles of given substance are connected by strong bonds, thus making it solid. Temperature will make the particles vibrate until they vibrate so hard they finally brake the bonds and the substance changes state from solid to liquid. In liquid state particles will still be connected by loose bonds and if we continue to heat up the given substance even more we will finally brake all the bonds and turn it to gas. Check out this interesting video to learn more.
